It, along with neon (Ne), is one of two elements that have never been observed to bond with another element in a compound. Helium is the second most abundant element in the universe and was discovered on the sun before it was found on Earth. What fun would a balloon be if it just laid on the floor? Stores use helium to fill up their balloons because it is less dense than air, which means it rises without being pushed. One group 18 element we're all familiar with is helium (He, Z = 2). Xenon glows blue when put in a vacuum tube and electricized. It is an odorless, colorless gas, but when it is put in a vacuum tube and excited with electricity it glows blue. Xenon was first called "the stranger" because it was the last noble gas to be isolated and characterized.

While most xenon is found as pure Xe, it can form complexes like XeF 6, XeF 4, XeO 3, and XePtF 6. One noble gas that exhibits some degree of reactivity is Xenon (Xe, Z = 54). They have very high ionization energies and negligible electronegativities because they have no desire to gain or lose an electron. The noble gases are happy with their completely filled valence shells.īecause of their uber-high stability, the noble gases are relatively unreactive.
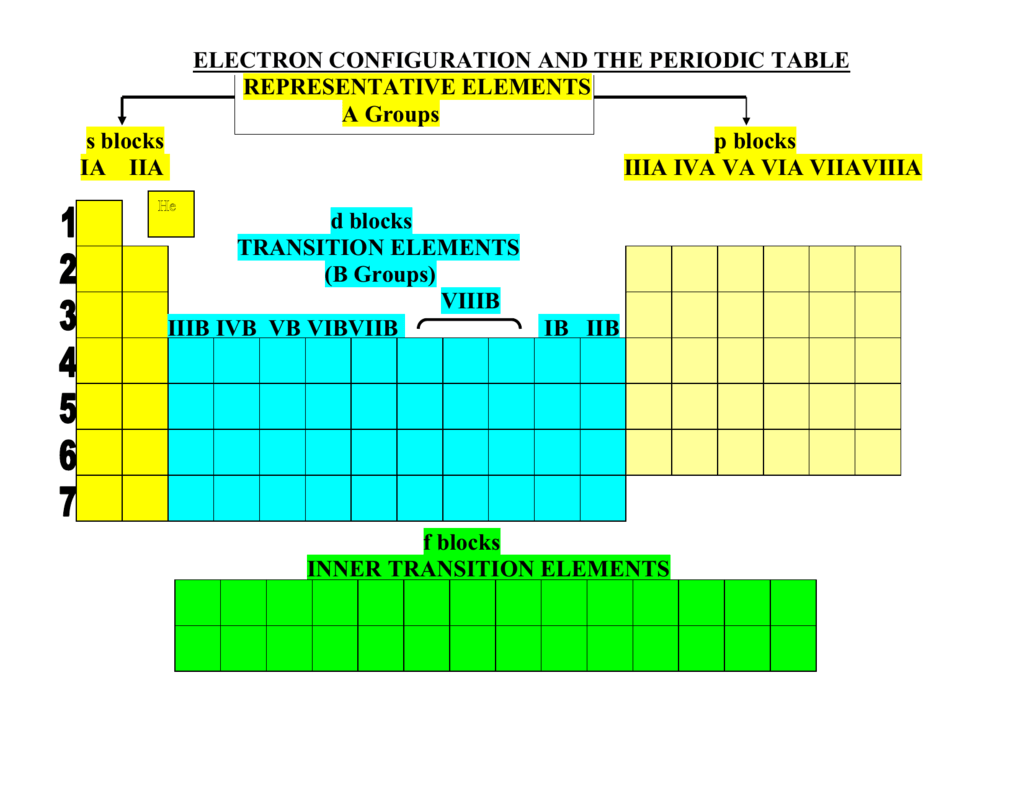
#Atom with ns2 electron configuration full
Helium is still happy because its outermost shell is completely full making it extremely stable. Even though it only has two electrons, it is grouped with elements that have eight valence electrons. It only has two electrons in its outer shell so its valence electron configuration is 1s 2. Helium is slightly different than the other noble gas elements. Two completely filled orbitals means ultimate stability. The valence electron configurations of these stable elements are: ns 2 np 6, where n is the period in which the gas resides. We've mentioned a gazillion times that other elements strive to achieve the stability of the noble gases, but where does this stability come from? Electrons. The noble part of their name actually arises from the German word edelgas, which means unreactive. These gases are not heir to the throne nor are they superior in morals. Collectively these gases are known as the noble gases. There are six gaseous elements located in group 8A (or group 18) of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn).

And now we've reached the last column of the p-block.
